Our Group organises 3000+ Global Events every year across USA, Europe & Asia with support from 1000 more scientific Societies and Publishes 700+ Open Access Journals which contains over 50000 eminent personalities, reputed scientists as editorial board members.
Open Access Journals gaining more Readers and Citations
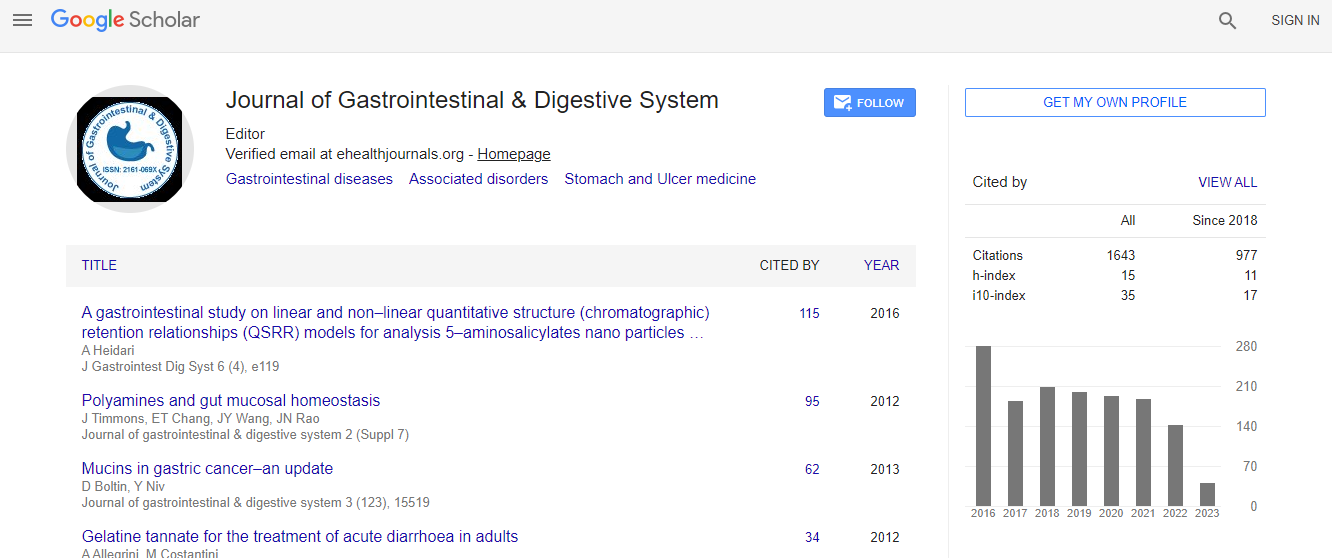
700 Journals and 15,000,000 Readers Each Journal is getting 25,000+ Readers
Citations : 2091
Indexed In
- Index Copernicus
- Google Scholar
- Sherpa Romeo
- Open J Gate
- Genamics JournalSeek
- China National Knowledge Infrastructure (CNKI)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- ICMJE
Useful Links
Recommended Journals
Related Subjects
Share This Page
HBsAg loss in HBeAg positive and HBeAg negative patients with chronic HBV treated with entecavir: A retrospective case series
11th Global Gastroenterologists Meeting
Tuzcuoglu I, Sungur M, Kurt K, Gokmen T and Ac�?±lar K
Merkez Efendi State Hospital, Turkey
Posters & Accepted Abstracts: J Gastrointest Dig Syst
DOI:
Abstract
We retrospectively investigated our patients who have been followed up in our gastroenterology and infectious diseases clinic between 2007 and 2016. All the patients were followed up at least six months before therapy to ensure that they had chronic hepatitis B. Every patient had liver biopsy procedure to assess the liver pathology. Of the patients who were started entecavir treatment, 161 patients had enrolled for this retrospective assessment. All the patients had continuous treatment (0.5 mg/day or 1 mg/day). Of these patients 30 were HBeAg positive (24 males, 6 females) and 129 HBeAg negative patients (99 males, 30 females) with chronic HBV infection, treatment initiated starting from 2007 till 2016. All the follow-ups for liver biochemistry were done every three months and HBV DNA was assessed every six months. HBsAg was controlled yearly. Total of nine patients had HBsAg loss (5.5%) (three patients of HBeAg+, and five patients of HBeAg-). Overall, the mean time to HBsAg loss was 3 years�?±4.5 months in HBeAg (+) patients and 3.5 years�?±7.5 months in HBe Ag (-) group. In this case series, HBsAg loss was observed both in HBeAg positive patients and in HBeAg negative patients. All of the patients with HBsAg loss received entecavir as 0.5 mg. Our results are consistent with the previous reports. Therefore, it may be suggested that treatment with entecavir could be associated to HBsAg loss in a period of time, in both HBeAg positive and HBeAg negative HBV patients.Biography
Email: isiltuzcu@yahoo.com